Erectile Dysfunction
Milton Lakin, MD
Hadley Wood, MD
Published: June 2018
Expire: June 2021
Since the 1990s, erectile dysfunction (ED) has been recognized as a common problem. Higher rates of reporting, diagnosis, and treatment of ED have been driven by effective treatments, direct to consumer advertising, and screening by health care providers.
Definition
Erectile dysfunction or disorder (ED) is the inability to develop and maintain an erection for satisfactory sexual intercourse or activity. Erectile dysfunction or erectile disorder are the preferred terms as opposed to impotence. There are no uniform criteria defining how consistent the problem has to be and for what duration it must be present to considered ED. The Diagnostic and Statistical Manual of Mental Disorder-5 specifies a duration of at least 6 months in its definition of ED.1
Prevalence
Several studies accessed the prevalence of ED. The Massachusetts Male Aging Study reported a prevalence of 52%.2 The study demonstrated that ED is increasingly prevalent with age: approximately 40% of men are affected at age 40 and nearly 70% of men are affected at age 70. The prevalence of complete ED increased from 5% at age 40 to 15% at age 70.2 Age was the variable most strongly associated with ED.
Incidence estimates have been published using data compiled from the Massachusetts Male Aging Study.3 Incidence data are necessary to assess risk and plan treatment and prevention strategies. The Massachusetts study data suggest there will be approximately 17,781 new cases of ED in Massachusetts and 617,715 in the United States annually.3
A larger national study, the National Health and Social Life Survey, looked at sexual function in men and women.4 This study surveyed 1,410 men aged 18 to 59 and also documented an increase in ED with age. Additionally, the study found a decrease in sexual desire with increasing age. Men in the oldest cohort (50 to 59) were more than 3 times as likely to experience erection problems and to report low sexual desire compared with men 18 to 29. Experience of sexual dysfunction was more likely among men in poor physical and emotional health. The study also concluded that sexual dysfunction is an important public health concern and that emotional issues are likely to contribute to the experience sexual dysfunction.
Pathophysiology
The development of an erection is a complex event involving integration of psychologic, neurologic, endocrine, vascular, and local anatomic systems. Positron emission tomography scanning studies have suggested that sexual arousal is activated in higher cortical centers that then stimulate the medial preoptic and paraventricular nuclei of the hypothalamus.5 These signals ultimately descend through a complex neural network involving the parasympathetic nervous system and eventually activate parasympathetic nerves in the sacral area (S2 to S4).
The neurovascular events that ultimately occur result in the inhibition of adrenergic tone and the release of the nonadrenergic, noncholinergic neurotransmitter, nitric oxide. Nitric oxide is believed to be released from nonadrenergic, noncholinergic nerves and endothelial cells. It subsequently stimulates the guanylate cyclase enzyme system in penile smooth muscle. This results in increased levels of cyclic guanosine monophosphate (cGMP) and ultimately in smooth muscle relaxation, enhancement of arterial inflow, and veno-occlusion, producing adequate firmness for sexual activity.
Recognized risk factors for ED include cardiovascular disease (CVD) (hypertension, atherosclerosis, and hyperlipidemia), diabetes, depression, alcohol use, smoking, pelvic/perineal surgery or trauma, neurologic disease, obesity, pelvic radiation, and Peyronie’s disease. One study suggested that the relationship between arterial disease and ED is very strong, with 49% (147 of 300) of patients with coronary artery disease noted on cardiac catheterization reporting significant erectile dysfunction.6 Endothelial dysfunction has been indicated as the pathophysiologic mechanism responsible for both CVD and ED.7 The Boston Area Community Health survey demonstrated a dose-response between smoking and incidence of erectile dysfunction.8 Animal studies have demonstrated both smooth-muscle disruption and decreased production of neural nitric oxide synthase in cigarette-exposed animals.9
Some evidence exists to suggest that chronic inflammation associated with metabolic syndrome also plays a role in endothelial dysfunction and erectile function, possibly due to oxidative stress.10
Examples of common neurologic conditions that can lead to ED include cerebral vascular accident, multiple sclerosis, Parkinson’s disease, and spinal cord injury. Microvascular disease associated with diabetes is thought to compound the endothelial and neural injuries associated with this disease. Pelvic surgery may disrupt both neural and vascular pathways, resulting in ED.
Hormone deficiency or hypogonadism, whether primary or secondary, has been thought to impact erectile function. Approximately a third of men in the European Male Aging Study demonstrated low testosterone, suggesting that hypogonadism is overrepresented among men with ED.11 Hormone deficiency, however, is less frequently the cause of ED than diabetes or vascular disease. Many entities with a strong relationship to ED also diminish bioavailable testosterone, including obesity, diabetes, and opioid use. Other hormones involved in testosterone metabolism or availability, like thyroid stimulating hormone and gonadotropins, also may impact erectile quality, presumably through regulating bioavailable testosterone. Understanding the relationship between testosterone and ED has been impaired by a lack of standardized measurement of this hormone and the cyclic nature of its release and consumption.
Signs and Symptoms
Some self-administered measures may be useful in the primary care setting to screen for and evaluate the degree of ED.12 The most commonly used instrument is the International Index of Erectile Function, a 15-item questionnaire that has been validated in many populations and is considered the gold standard to evaluate patients for ED.13 The Sexual Health Inventory for Men is a short-form, 5-item questionnaire developed to monitor treatment progress.12 It is important to recognize that short-form questionnaire does not evaluate specific areas of the sexual cycle, such as sexual desire, ejaculation, and orgasm; however, it may be useful in discussing ED with patients and evaluating treatment results over time.
Diagnosis
If it is determined that ED is a problem, the patient evaluation should include a detailed sexual and medical history and a physical exam. In particular, it is important to evaluate the ED within the context of ejaculatory problems. There is a strong interplay between premature ejaculation (PE) and ED, with about a third of ED patients reporting PE. The relationship between the PE and ED is bidirectional and successful treatment of one often requires treatment of the other.14
A number of specific questions relating to sexual function can help the clinician evaluate the complaint of ED. Questions should focus on the following:
- How long has ED been a problem, and did it start gradually or suddenly?
- How frequently do you have intercourse currently, and how frequent was it in the past? Do you have difficulty with penetration and/or loss of the erection during intercourse in the absence of premature ejaculation?
- How firm are your erections (use a scale of 1 to 10)? Do your erections vary under different circumstances, such as with different partners, oral stimulation, or masturbation?
- Do you have morning or evening erections and, if so, what is the quality of these erections?
- Is there any new curve or bend to your penis to suggest Peyronie’s disease? If curvature is present, is it painful? What are the location and severity of the curvature?
- Are you having any difficulties with sexual desire, arousal, ejaculation, or orgasm? If so, did these difficulties occur with the onset of the erectile dysfunction or are they separate issues?
Once questions related to the specific erectile complaint have been reviewed, additional questions relating to medical history and psychosocial factors need to be evaluated. In particular, these include the following:
- symptoms suggesting the presence of diabetes, peripheral vascular disease, neurologic disease, or chronic liver or kidney disease;
- a complete list of medications and recreational drugs, including over the counter “prostate supplements” which may have anti-androgen effects and questions about cigarette smoking;
- history of surgery or radiation therapy, particularly procedures related to genitourinary or gastrointestinal malignancy;
- a history of pelvic genital, perineal, or spinal cord trauma;
- the quality of the marital or partner relationship and expectations of both patient and partner.
Following a review of the medical history, the salient features of the physical examination should include the following:
- An assessment of the patient's general health and affect, as well as secondary sexual characteristics, noting in particular gynecomastia and hair loss (axillary or pubic).
- Careful peripheral vascular examination that includes palpation of the lower extremity pulses as well as auscultation for bruits in the abdominal and femoral regions.
- Detailed neurologic examination to include gait and postural instability, with blood pressure changes, distal extremity and saddle sensation, and reflexes, including cremasterics and bulbocavernosus.
- Careful genital examination, noting testicular size (to screen for hypogonadism) and palpating the penis for Peyronie’s plaques.
- Rectal examination to assess sphincter tone and evaluate the prostate.
- Careful abdominal examination looking for organomegaly masses or other signs of liver or kidney disease.
- Cardiopulmonary examination to help evaluate the patient's fitness for future treatment options.
Once a complete sexual and medical history has been completed, appropriate laboratory studies should be conducted. In the initial evaluation of ED, sophisticated laboratory testing is rarely necessary. For example, serum testosterone (and sometimes prolactin) is typically only useful when the patient demonstrates hypogonadal features or testicular atrophy, or when clinical history is suggestive. Additional hormonal evaluation may include thyroid stimulating hormone in those with a clinical suspicion of hypothyroidism or appropriate diabetes screening in those presenting with a concern for impaired glucose metabolism. If the patient has not been evaluated with a lipid panel and hyperlipidemia is suspected, measurement and appropriate referral to internal medicine or cardiology is recommended. In most cases, a tentative diagnosis can be established with a complete sexual and medical history, physical examination, and limited or no laboratory testing.
Treatment
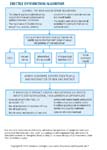
The 2018 American Urological Association Guidelines for the treatment of Erectile Dysfunction recommend a complete history and physical and lifestyle modifications followed by a shared-decision-making approach for the existing medical treatments (Figure 1).15
Lifestyle Modification
Erectile dysfunction is known to be associated with general health status, thus, lifestyle modification improves erectile function and decreases the rate of decline of function with aging. One year after discontinuation of smoking, patients were found to have a 25% improvement in erectile quality.16 In addition, multivariate analysis found obesity is associated with erectile dysfunction with an approximately 50% increase in ED in obese men as compared with normal weight men.17
Over a 2-year period, a third of the men randomized to a weight loss program demonstrated resolution of erectile dysfunction.10 A Mediterranean diet and nutritional counseling reported increased erectile quality.18 Little evidence supports that increased physical activity alone improves erectile quality; however, the strong association between physical activity and lower BMI is well described, and therefore recommended for men with erectile dysfunction and without a contraindication to physical activity.
The Massachusetts Male Aging Study demonstrated increased risk of ED among heavy alcohol users though the impact of alcohol use on erection quality is not well understood.2
Oral Medications
Although there are numerous options for nonsurgical treatment, oral phosphodiesterase-5 (PDE5) inhibitors should be offered as first-line treatment of patients with ED unless contraindicated.19 Table 1 summaries the oral PDE5 drugs approved by the U.S. Food and Drug Administration (FDA) for ED.
Table 1: Phosphodiesterase-5 inhibitors for erectile dysfunction
| Generic (Brand) | Median tmax, minutes | Half-life, hours | Absorption effected by food | Dosed | Side effects |
|---|---|---|---|---|---|
| Avanafil (Stendra) | 30 to 45 |
5 to 10 | No | as needed | Decreased blood pressure, headache, flushing (12%-16%); nasal congestion (2%-4%); gastric reflux, nausea (5%-7%); priapism (very rare); leg-buttock pain (vardenafil, tadalafil) |
| Sildenafil (Viagra) | 60 |
3 to 5 | Yes (high fat food) | as needed | |
| Vardenafil (Levitra) | 60 |
4 to 5 | Yes (high fat food) | as needed | |
| Tadalafil (Cialis) | 120 |
17.5 | No | daily or weekender |
tmax = time to maximum serum concentration
Data from references 20-23.
These oral medications reversibly inhibit penile-specific PDE5 and enhance the nitric oxide–cGMP pathways of cavernous smooth muscle relaxation; that is, all prevent the breakdown of cGMP by PDE5. It is important to emphasize to patients that these drugs augment the body’s natural erectile mechanisms, therefore the neural and psychoemotional stimuli typically needed for arousal still need to be activated for the drugs to be efficacious.
Efficiacy
According to a review of all randomized controlled trials evaluating sildenafil by the American Urological Association (AUA) Consensus Panel on Erectile Dysfunction, 36% to 76% of patients receiving the drug were "able to achieve intercourse" during treatment. For tadalafil, four randomized controlled trials revealed that 11% to 47% of patients were "able to achieve intercourse." Similar efficacy has been observed with vardenafil, although studies are fewer.19 A meta-analysis published in 2013 clearly demonstrated increased efficacy over placebo for all PDE5 inhibitors.24 Head-to-head comparison suggested that tadalafil outperforms sildenafil on validated measures of erectile dysfunction, including the international index of erectile function and sexual encounter profile-2 and -3.
Side effects
Oral ED drugs are generally well tolerated. Typical side effects include headache, flushing, dyspepsia, and nasal congestion. Visual abnormalities are encountered with sildenafil, but are unlikely with vardenafil and tadalafil. Back pain and myalgia can occur with tadalafil, but are unusual with sildenafil or vardenafil.
Contraindications
Patients at high cardiovascular risk should not be treated for ED until their cardiac condition is stabilize. These conditions include unstable or refractory angina, myocardial infarction or cerebrovascular accident within the past 2 weeks, uncontrolled hypertension, New York Heart Association (NYHA) Functional Classification III-IV congestive heart failure, high-risk arrhythmias, hypertrophic obstructive cardiomyopathies, and moderate-to-severe valvular disease.25 This class of drugs is also contraindicated in patients who use nitroglycerin or nitrate-containing compounds.26, 27
Although vardenafil does not seem to produce significant clinical QT prolongation, it has been suggested that it be avoided in patients who have congenital QT prolongation abnormalities and in patients using class I antiarrhythmic drugs, such as quinidine and procainamide. It is also best to avoid the use of vardenafil with class III antiarrhythmic drugs, such as amiodarone or sotalol.
Due to the risk of hypotension, caution should be used in patients using alpha blockers for prostate hyperplasia and patients using other antihypertensive medications and alpha blockers, which should not be co-administered with PDE5 inhibitors. In patients who take 50 mg of sildenafil or more and use alpha blockers, sildenafil dosing should be avoided for at least 4 hours after the dose of the alpha blocker. In patients who take 25 mg of sildenafil, use of any alpha blockers is considered safe.
Dose-adjustment is recommended in patients with hepatic failure and with co-administration of drugs also metabolized by the cytochrome p450 enzymes.
There is a reported risk for nonarteritic anterior ischemic optic neuropathy (NAION) with the use of PDE5 inhibitors. Although the association is controversial, it is prudent to consider alternative treatments for ED in patients with NAION or at risk for it.28
Withdrawal of Medications Causing or Contributing to ED
It is extremely important to take a complete drug history for patients reporting ED. Medications that may cause or contribute to ED include:
- Alcohol, nicotine, and illicit drugs
- Analgesics
- Anticonvulsants
- Antidepressants
- Antihistamines
- Antihypertensives
- Antiparkinson
- Antipsychotics
- Cardiovascular agents
- Cytotoxic agents
- Diuretics
- Hormones and hormone-active agents
- Immunomodulators
- Tranquilizers.29
Of particularly concern are antihypertensive medications for CVD (eg, digoxin, disopyramide [Norpace], gemfibrozil [Lopid]), anxiety, depression (eg, lithium, monoamine oxidase inhibitors, selective serotonin reuptake inhibitors, serotonin-norepinephrine reuptake inhibitors, tricyclic antidepressants), or psychosis (eg, chlorpromazine, haloperidol, pimozide [Orap], thioridazine, thiothixene). Antihypertensive drugs, such as diuretics (eg, spironolactone, thiazides) and beta blockers, may be associated with ED. Discontinuation or switching to alternative drugs, such as angiotensin-converting enzyme inhibitors or calcium channel blockers (eg, diltiazem, nifedipine, amlodipine), may reduce ED. The newer angiotensin II receptor antagonists may be less problematic with respect to ED, but long-term data is needed to evaluate this.
Of the drugs used for depression, tricyclic antidepressants may be associated with erectile problems and other drugs may be substituted to prevent this complication. Currently available substitutes include bupropion, nefazodone, and trazodone. The selective serotonin reuptake inhibitors (eg, fluoxetine, sertraline, paroxetine, citalopram) can also cause difficulties with ED, but they might also have other significant sexual side effects, including decreased libido and anorgasmia.
Clinical experience in switching medications to improve ED has been disappointing in that improvement does not often occur. Nonetheless, it is important to try to discontinue possible offending medications before proceeding to more invasive ED treatment options. Oral ED medications have changed the way clinicians discontinue medications in patients with ED and has improved the approach. For example, a patient may develop ED on a thiazide diuretic. The diuretic may be withdrawn, but a trial of oral ED therapy can be initiated during the observation period while the patient is waiting to see if any spontaneous improvement in ED occurs after drug withdrawal. Alternatively, if diuretic therapy is effective, well tolerated, and controlling blood pressure, oral ED therapy can be used on an ongoing basis to treat the side effect of ED.
If a trial of oral ED therapy and withdrawal of offending medications prove to be ineffective in restoring erectile function, it is appropriate for most primary care practitioners to consider referral to a specialist for additional evaluation and discussion of alternative treatment options. These include intracavernous injection therapy, vacuum constriction devices, intraurethral therapy, and possible surgery.
Vacuum Constriction Devices
If a trial of oral therapy and withdrawal of offending medications do not restore erectile function or if a patient has medical or financial contraindications to pharmacologic therapy, most primary care practitioners should consider referring the patient to a specialist for additional evaluation and discussion of alternative treatment options. However, some primary care practitioners may recommend vacuum constriction devices.
The device consists of an acrylic cylinder placed over the penis that uses a lubricant to achieve a good seal between the penile body and cylinder. An erection is then achieved by creating a vacuum inside the cylinder with a pump connected to the cylinder. Once an erection is achieved, a constriction band is applied to the base of the penis to maintain the erection. The cylinder can then be removed and the patient can engage in intercourse with the constriction band at the base of the penis maintaining the erection. The band can remain on for approximately 30 minutes and then must be removed. The erection produced by the device differs from a normal erection likely because of venous occlusion from the constriction band resulting in generalized swelling of the entire penis, with probable preservation of arterial inflow.
Clinical studies have suggested that these devices are effective and acceptable to a large number of patients with ED of varying causes, including psychogenic erectile failure. These devices are safe and can restore a man’s ability to achieve penetrative intercourse, with one study suggesting nearly 95% success with adequate instruction and support.30 However, satisfaction with this treatment modality typically wanes with time, as patients report dissatisfaction with how cumbersome or unnatural the devices are to use, hinging or buckling of the erection with thrusting, and dissatisfaction with the fact that the erection is ischemic and therefore cold, which can be off-putting to the partner.
There are relatively few contraindications to the use of vacuum devices. Some conditions can predispose to priapism or perhaps bleeding with constriction, such as sickle cell disease, polycythemia, and other blood dyscrasias. Patients taking anticoagulants can safely use vacuum constriction devices but need to accept a higher risk of bleeding (ecchymosis). Good manual dexterity is also needed to use the device; if manual dexterity is impaired, a willing sexual partner can learn to apply the device.
Complications from the use of a vacuum constriction device are relatively minor. They include the development of petechiae or ecchymosis, numbness or coolness of the penis, trapping of the ejaculate, and pivoting of the penis at the base.
Intraurethral and Intracorporeal Alprostadil
Alprostadil (also known as prostaglandin E1 [PGE1]) is the prominent known smooth-muscle dilator of the corpus cavernosum. Its mechanism of action is believed to be the promotion of intracellular accumulation of cyclic adenosine monophosphate, thereby causing decreased intracellular accumulation of calcium and resulting smooth muscle relaxation. Alprostadil can be delivered to the erectile tissue either via an intraurethral suppository that is massaged and then absorbed across the corpus spongiosum of the urethra to the corpora cavernosa, or directly injected into the corpora cavernosa. When administered urethrally, doses are substantially higher than when directly injected (typical dosing is 500 mcg to 1 mg intraurethral compared with 2.5 mcg to 20 mcg intracavernosal).
Side effects include lightheadedness, fainting, priapism, urethral bleeding (intraurethral), dyspareunia in the partner (intraurethral), hematoma (intracavernosal) or penile curvature secondary to scar (intracavernosal). Efficacy of intraurethral alprostadil has been demonstrated to be around 50% ("able to have intercourse") in randomized controlled trials.31,32 For intracorporeal injection, typically alprostadil is tried alone, or compounded with papaverine (nonspecific phosphodiesterase inhibitor that increases intracellular cyclic adenosine monophosphate and cGMP) and/or phentolamine (competitive, non-selective alpha1- and alpha2-adrenoreceptor blocker). Pinsky et al33 reported an extensive review of the benefits and drawbacks of the combinations of these drugs.
Given the high risk of priapism during escalation of therapy for intracorporeal injection, it is recommended that the drugs be administered in a supervised office visit initially and that the patient be given a well-articulated plan for treatment of priapism if it occurs. Escalation guidelines for alprostadil alone vary, but a general guideline is to start at 2.5 mcg and increase by 2.5 mcg to a dose of 5 mcg and then in increments of 5 mcg to 10 mcg until an erection sufficient for penetration, not lasting more than 1 hour, is achieved. If there is no response to the initial 2.5-mcg dose, escalation dosing can be slightly more liberal.34 A European prospective trial of PGE1 alone found 91% of the 54 patients completing the 4 years of the study reported good or better tolerability and satisfaction with therapy.35
Other Nonsurgical Treatments for Erectile Dysfunction
Several treatments were promoted in the pre-PGE1, pre-prostaglandin era, including yohimbine, trazodone, testosterone, and various herbal remedies. None of these is currently recommended under the updated American Urological Association Guidelines for the Treatment of Erectile Dysfunction.15 Testosterone supplementation is only recommended for men with low testosterone levels.
Surgical Treatments for Erectile Dysfunction
Implantation of penile prosthesis remains an important option for men with ED if medical treatment fails or is inappropriate. Prostheses are available as a saline-filled silicone device or a malleable device. The benefit of the former is a more natural appearance in the deflated state, closely approximating the appearance of a flaccid penis. The trade-off is a higher mechanical failure rate and higher cost. Satisfaction rates for patients who underwent penile prosthesis surgery have been reported to be near 90%.36 However, in the majority of patients who receive this treatment, less invasive alternatives have failed and therefore satisfaction with this treatment would be expected to be higher in this subset of patients. Risks of these devices include surgical and anesthetic risk, device infection, and device malfunction. Mechanical failure rates depend on the specific device being investigated. Overall, the percentage of devices that are free from mechanical failure at 5 years ranges from 84% to 94%.19 Infection rates in the era of coated devices and improved techniques are reported to be less than 1%.37
All devices that are currently approved by the FDA are considered safe for use in magnetic resonance imaging environments. However, 2 previously approved devices–the OmniPhase and the DuraPhase penile prostheses–are not considered safe in this environment. Other surgical procedures–including venous ligation to limit penile venous outflow and penile revascularization procedures–are rarely successful and are not recommended.19 These surgeries are only indicated when a patient demonstrates recent-onset ED and an occlusive lesion seen on angiogram or magnetic resonance angiography and should be performed only in centers of excellence for ED.
Summary
- Erectile dysfunction (ED) is the inability to develop and maintain an erection for satisfactory sexual intercourse.
- Risk factors for ED include diabetes mellitus, increasing age, dyslipidemia, hypertension, and use of antihypertensive and psychotropic medications.
- Proper evaluation includes a careful history, physical examination, and evaluation for possible endocrine causes (eg, low testosterone, high prolactin levels).
- Because erectile dysfunction is caused by a complex set of psychosocial, neurologic, and vascular factors, a specific cause in a patient may remain ambiguous.
- Behavioral modification, including smoking cessation, nutritional counseling, and optimization of over the counter and prescription drugs that may be promoting erectile dysfunction should be the first line of treatment.
- Oral therapy with PDE5 inhibitors is often successful and safe for appropriately selected patients and should be considered the first line of treatment.
- Vacuum constriction devices provide acceptable alternative therapy for patients who do not qualify for medical treatment.
- Alternative therapies, including intracorporeal injection and intraurethral suppositories of prostaglandins should be administered selectively.
- Surgical implantation of a penile prosthesis represents the only well-accepted surgical therapy for the treatment of ED.
References
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 5th ed. Washington, DC: American Psychiatric Association; 2013.
- Feldman HA, Goldstein I, Hatzichristou DG, Krane RJ, McKinlay JB. Impotence and its medical and psychosocial correlates: Results of the Massachusetts Male Aging Study. J Urol 1994; 151:54–61.
- Johannes CB, Araujo AB, Feldman HA, Derby CA, Kleinman KP, McKinlay JB. Incidence of erectile dysfunction in men 40 to 69 years old: Longitudinal results from the Massachusetts Male Aging Study. J Urol 2000; 163:460–463.
- Laumann EO, Paik A, Rosen RC. Sexual dysfunction in the United States: Prevalence and predictors. JAMA 1999; 281:537–544.
- Goldstein I. Male sexual circuitry. Working group for the study of central mechanisms in erectile dysfunction. Sci Am 2000; 283:70–75.
- Montorsi F, Briganti A, Salonia A, et al. Erectile dysfunction prevalence, time of onset and association with risk factors in 300 consecutive patients with acute chest pain and angiographically documented coronary artery disease. Eur Urol 2003; 44:360–364; discussion 364–365.
- Guay AT. Relation of endothelial cell function to erectile dysfunction: Implications for treatment. Am J Cardiol 2005; 96:52M–56M.
- Kupelian V, Link CL, McKinlay JB. Association between smoking, passive smoking, and erectile dysfunction: Results from the Boston Area Community Health (BACH) Survey. Eur Urol 2007; 52:416–422.
- Imamura M, Waseda Y, Marinova GV, et al. Alterations of NOS, arginase, and DDAH protein expression in rabbit cavernous tissue after administration of cigarette smoke extract. Am J Physiol Regul Integr Comp Physiol 2007; 293:R2081–2089.
- Yudkin JS, Stehouwer CD, Emeis JJ, Coppack SW. C-reactive protein in healthy subjects: Associations with obesity, insulin resistance, and endothelial dysfunction: A potential role for cytokines originating from adipose tissue? Arterioscler Thromb Vasc Biol 1999; 19:972–978.
- Wu FC, Tajar A, Beynon JM, et al; EMAS Group. Identification of late-onset hypogonadism in middle-aged and elderly men. N Engl J Med 2010; 363:123–135.
- Rosen RC, Cappelleri JC, Smith MD, et al: Development and evaluation of an abridged 5-item version of the International Index of Erectile Function (IIEF 5) as a diagnostic tool for erectile dysfunction. Int J Impot Res 1999; 11:319–326.
- Rosen RC, Riley A, Wagner G, Osterloh IH, Kirkpatrick J, Mishra A. The International Index of Erectile Function (IIEF): A multidimensional scale for assessment of erectile dysfunction. Urology 1997; 49:822–830.
- Corona G, Rastrelli G, Limoncin E, Sforza A, Jannini EA, Maggi M. Interplay between premature ejaculation and erectile dysfunction: A systematic review and meta-analysis. J Sex Med 2015; 12:2291–2300.
- Burnett AL, Nehra A, Breau RH, et al. Erectile dysfunction: AUA guideline (2018). American Urological Association website. https://www.auanet.org/guidelines/erectile-dysfunction-aua-guideline-(2018). Published 2018. Accessed June 6, 2018.
- Pourmand G, Alidaee MR, Rasuli S, Maleki A, Mehrsai A. Do cigarette smokers with erectile dysfunction benefit from stopping?: A prospective study. BJU Int 2004; 94:1310–1313).
- Janiszewski PM, Janssen I, Ross R. Abdominal obesity and physical inactivity are associated with erectile dysfunction independent of body mass index. J Sex Med 2009; 6:1990–1998.
- Esposito K, Ciotola M, Giugliano F, De Sio M, Giugliano G, D’armiento M, Giugliano D. Mediterranean diet improves erectile function in subjects with the metabolic syndrome. Int J Impot Res 2006; 18:405–410.
- Montague DK, Jarow JP, Broderick GA, et al; Erectile Dysfunction Guideline Update Panel. Chapter 1: The management of Erectile dysfunction: An AUA update. J Urol 2005; 174:230–239.
- Avanafil (Stendra) package insert
- Sildenafil (Viagra) package insert
- Vardenafil (Levitra) package insert
- Tadalafil (Cialis) package insert
- Yuan J, Zhang R, Yang Z. Comparative effectiveness and safety of oral phosphodiesterase type 5 inhibitors for erectile dysfunction: a systematic review and network meta-analysis. Eur Urol 2013; 63:902–912.
- Jackson G, Rosen RC, Kloner RA, Kostis JB. The second Princeton consensus on sexual dysfunction and cardiac risk: New guidelines for sexual medicine. J Sex Med 2006; 3:28–36.
- DeBusk R, Drory Y, Goldstein I, et al: Management of sexual dysfunction in patients with cardiovascular disease: Recommendations of the Princeton Consensus Panel. Am J Cardiol 2000; 86:175–181.
- DeBusk RF, Pepine CJ, Glasser DB, et al: Efficacy and safety of sildenafil citrate in men with erectile dysfunction and stable coronary artery disease. Am J Cardiol 2004; 93:147–153.
- Pomeranz HD Erectile dysfunction agents and nonarteritic anterior ischemic optic neuropathy. Neurol Clin 2017; 35:17–27.
- Rew KT, Heidelbaugh JJ. Erectile dysfunction. Am Fam Physician 2016; 94:820–827.
- Khayyamfar F, Forootan SK, Ghasemi H, Miri SR, Farhadi E. Evaluating the efficacy of vacuum constrictive device and causes of its failure in impotent patients. Urol J 2014; 10:1072–1078.
- Padma-Nathan H, Hellstrom WJ, Kaiser FE, et al: Treatment of men with erectile dysfunction with transurethral alprostadil. Medicated Urethral System for Erection (MUSE) Study Group. N Engl J Med 1997; 336:1–7.
- Williams G, Abbou CC, Amar ET, et al: Efficacy and safety of transurethral alprostadil therapy in men with erectile dysfunction. MUSE Study Group. Br J Urol 1998; 81:889–894.
- Pinsky MR, Chawla A, Hellstrom WJG. Intracavernosal therapy and vacuum devices to treat erectile dysfunction. Arch. Esp. Urol 2010; 63: 717–725.
- Linet OI, Ogrinc FG. Efficacy and safety of intracavernosal alprostadil in men with erectile dysfunction. The Alprostadil Study Group. N Engl J Med 1996; 334:873–877.
- Porst H, Buvat J, Meuleman E, Michal V, Wagner G. Intracavernous alprostadil alfadex--an effective and well tolerated treatment for erectile dysfunction. Results of a long-term European study. Int J Impot Res 1998; 10:225–231.
- Ji YS, Ko YH, Song PH, Moon KH. Long-term survival and patient satisfaction with inflatable penile prosthesis for the treatment of erectile dysfunction. Korean Journal of Urology 2015; 56:461-465.
- Eid JF, Wilson SK, Cleves M, Salem EA. Coated implants and "no touch" surgical technique decreases risk of infection in inflatable penile prosthesis implantation to 0.46%. Urology 2012; 79:1310–1315.