Pregabalin (Lyrica®):Part I
Volume VIII, Number 5 | September/October 2005
Chadrick Lowther, Pharm.D.
Return to Pharmacotherapy Update Index
Introduction
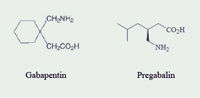
Pregabalin (Lyrica®; Pfizer) is classified as a miscellaneous analgesic and anticonvulsant.1-4 Pregabalin received Food and Drug Administration (FDA) approval on December 30, 2004, for the management of neuropathic pain associated with diabetic peripheral neuropathy (DPN) and postherpetic neuralgia (PHN); moreover, pregabalin is approved for use as adjunctive therapy for adult patients with partial onset seizures.3,5 Pregabalin is structurally related to gabapentin (Neurontin®; Pfizer) (See Figure 1).4,6
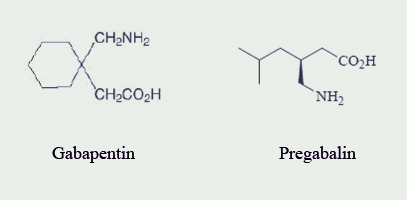
Gabapentin was approved by the FDA on December 30, 1993, for adjunctive therapy in the treatment of partial seizures in adults and pediatric patients (3-12 years of age) and also approved in 2002 for pain management of PHN in adults.6 However, during the past 12 years, off-label use has accounted for the largest percentage of gabapentin prescriptions.7 Some of these off-label uses include: panic disorder, migraine prophylaxis, social phobia, mania, bipolar disorder, and alcohol withdrawal.8 Gabapentin lost its product exclusivity on May 24, 2005, and subsequently Pfizer introduced pregabalin with an FDA-approved indication for DPN (See Table 1).5,7
Table 1: FDA-Approved Indications for Pregabalin and Gabapentin
| Indications | Pregabalin | Gabapentin |
|---|---|---|
| Neuropathic pain associated with diabetic peripheral neuropathy | x | |
| Neuropathic pain associated with postherpetic neuralgia | x | x |
| Adjunctive therapy for adults with partial onset seizures | x | |
| Adjunctive therapy for partial seizures with or without secondary generalization in patients older than 12 years of age with epilepsy | x | |
| Adjunctive therapy in partial seizures in pediatric patients 3-12 years of age | x |
This article will consist of two parts. Part I focuses on pharmacology, pharmacokinetics, and selected clinical trials. Part II will review warnings and precautions, adverse reactions, drug interactions, and dosing.
Pharmacology
Pregabalin is a structural derivative of the inhibitory neurotransmitter gamma-aminobutyric acid (GABA); however, it does not directly bind to GABA receptors. Pregabalin binds to the alpha2-delta site of voltage-gated calcium channels in the central nervous system (CNS) tissues.4
This calcium channel modulation may reduce the release of many neurotransmitters.4
Like gabapentin, the precise mechanism of action of pregabalin is unknown.4 However, gabapentin differs from pregabalin because of the reduced binding affinity to voltage-gated calcium channels. 6 Pregabalin is six-times more potent than gabapentin in binding affinity to the alpha2-delta voltage-gated calcium channel.9 The manufacturer states that 50 mg of pregabalin is approximately equal to 300 mg of gabapentin. This alteration of calcium channel function is not to be confused with calcium-channel blockers. Pregabalin and gabapentin alter channel function without complete blockade of the calcium channel resulting in virtually no change in systemic blood pressure or coronary blood flow changes.9
Pharmacokinetics
The peak oral concentration of pregabalin occurs after 1.5 hours with bioavailability being greater than 90% and independent of the dose. Pregabalin can be administered with food since it does not have a clinical effect on total absorption; however, food does decrease the peak plasma concentration by 25% to 30% and increases the time-to-peak plasma concentration to approximately 3 hours.4 Pregabalin does not bind to plasma proteins and is eliminated primarily by the kidneys as unchanged drug; furthermore, the half-life is approximately 6.3 hours.4 The elimination of pregabalin is nearly proportional to creatinine clearance; therefore, renal dose adjustment is necessary in renal dysfunction.4 Additionally, pregabalin is effectively removed by hemodialysis.4
Gabapentin displays variable bioavailability when being administered with or without food, and bioavailability decreases with increases in the dose.6 Food slightly increases the rate and extent of gabapentin absorption.6 The half-life of gabapentin is 5 to 7 hours, and it is eliminated unchanged in the urine. Due to the renal clearance of gabapentin, dose adjustments are required for renal dysfunction.6
Selected Clinical Trials
The following clinical trials examine the FDA-approved indications of pregabalin. To date, there are no direct comparative, randomized clinical trials evaluating the differences in efficacy and safety between pregabalin and gabapentin in adults or children. However, there have been pharmacology studies as well as animal models that have recognized the comparable binding sites of voltage-gated calcium channels as well as the increased binding activity of pregabalin versus gabapentin.10-12
Diabetic Peripheral Neuropathy
Individuals with uncontrolled blood glucose values can experience diabetes associated nerve pain, known as diabetic peripheral neuropathy (DPN). It is often described as sensations of burning, shooting, and tingling. Elevated blood glucose levels can cause damage to nerves, muscles, and vascular tissue in the feet, legs, arms, and hands. As evaluated by the Diabetes Control and Complications Trial (DCCT), strict blood glucose control decreases the incidence and progression of DPN.13 Additionally, DPN results in substantial morbidity including an increased number of recurrent lower extremity infections, ulcerations, and subsequent amputations.14 Clinical trials have shown pregabalin to be effective in the elimination of symptoms associated with DPN.15,16
Lesser and colleagues compared pregabalin with placebo in a randomized, double-blind, 5-week study including 338 patients with a 1- to 5-year history of painful DPN.15 At enrollment, patients had an average weekly pain score of 4 or greater on an 11-point numeric pain-rating scale (0 = no pain; 10 = worst possible pain). Failure to respond to gabapentin at doses > 1200 mg/day for pain associated with DPN was an exclusion criterion of the study. Patients were randomized to treatment with pregabalin 75 mg/day (n=77), pregabalin 300 mg/day (n=81), pregabalin 600 mg/day (n=82), or placebo (n=97) for 5 weeks. Doses were administered three times daily. Pregabalin doses were titrated over 6 days in the 600 mg/day group. The primary efficacy endpoint was the change in the mean pain score recorded in the patients' daily pain diaries. The mean change in the pain score was -1.26 with pregabalin 300 mg/day (95% CI, -1.86, -0.65; p= 0.0001) and -1.45 with pregabalin 600 mg/day (95% CI, -2.06, -0.85; p = 0.0001), compared with placebo. These two doses were also associated with significant improvements in the Short Form-McGill Pain Questionnaire (SF-MPQ) total score, sleep interference score, Visual Analog Scale (VAS) score, and Present Pain Index (PPI) score, beginning at week 1 and continuing throughout the remainder of the study. The lowest pregabalin dosage (75 mg/day) was no better than placebo in any of the outcome parameters. A 50% or greater reduction in pain, compared with baseline, was achieved in 46% of patients in the pregabalin 300 mg/day group, 48% in the pregabalin 600 mg/day group, and 18% in the placebo group. The authors concluded that the efficacy profile for pregabalin was both statistically and clinically significant and is in the same range as other drugs of first choice currently used to treat neuropathic pain associated with DPN. Additionally, pregabalin was safe and well-tolerated.
Rosenstock and colleagues conducted a multicenter, randomized, double-blind, parallel-group, 8-week comparison trial to evaluate the effectiveness of pregabalin (n=76) compared to placebo (n=70) in alleviating pain in patients with a 1-to 5-year history of painful DPN.16 Failure to respond to gabapentin at doses > 1200 mg/day for pain associated with DPN was an exclusion criterion of the study. After the 1-week baseline phase, the 8-week fixed-dose, double-blind treatment phase with pregabalin 100 mg three times a day or placebo was initiated. The primary efficacy parameter was mean pain score from diary entries. In addition, there were six secondary endpoints to assess efficacy including SF-MPQ, daily sleep interference score, Patient Global Impression of Change (PGIC), Clinical Global Impression of Change (CGIC), SF-36 Health Survey, and the Profile of Mood States (POMS). The mean change in the pain score was -1.47 with pregabalin 300 mg/day (95% CI, -2.19, -0.75; p = 0.0001) compared to placebo. Pregabalin also showed statistically significant improvements versus placebo in mean sleep interference scores, total SF-MPQ score, SF-36 Bodily Pain subscale, PGIC, and Total Mood Disturbance and Tension–Anxiety components of POMS. A 50% or greater reduction in pain, compared with baseline, was achieved in 40% of patients in the pregabalin 300 mg/day group and 14.5% in the placebo group (p=0.001). Dizziness, somnolence, infection (colds and upper respiratory tract infections), and peripheral edema occurred more frequently in pregabalin-treated patients compared to placebo-treated patients. The authors concluded that pregabalin is effective and rapid in decreasing the neuropathic pain associated with DPN in patients with diabetes reporting moderate-to-severe chronic pain. Furthermore, it was safe and well-tolerated.
Postherpetic Neuralgia
Postherpetic neuralgia (PHN) is traditionally defined as the persistence of pain for more than 1 month after the disappearance of the rash associated with shingles.17 Shingles is caused by the Varicella-zoster virus (VZV) and can persist for years in the dorsal root ganglia of cranial or spinal nerves after resolution of the original infection. Activation of VZV can be due to an immunocompromised status causing an acute neuritis.18,19 Many patients may continue to experience pain for months to years after the resolution of the rash and management can be challenging.18,19 A number of different medications have been used for PHN such as, antiviral agents [e.g., famciclovir (Famvir®) or valacyclovir (Valcyte®)], prednisone, lidocaine patches, amitriptyline, and gabapentin.20 Clinical trials have shown a reduction in PHN with the use of pregabalin.21,22
Dworkin and colleagues performed an 8-week multicenter, randomized, double-blind, parallel group study to evaluate the efficacy and safety of pregabalin in the treatment of PHN (n=173).22 Eligible patients included those > 18 years of age, had pain defined as the presence of pain for more than 3 months after healing of a herpes zoster skin rash, and that pain was at least 40 mm on the 100 mm on the visual analog scale from the SF-MPQ. Patients currently receiving gabapentin were required to stop the drug at least 7 days prior to study medication. Failure to respond to gabapentin at doses > 1200 mg/day for pain associated with DPN was an exclusion criterion of the study. Patients were randomized to either placebo or pregabalin 50 mg three times a day for the first 3 days and 100 mg three times a day for the remainder of the first week. During the second week, those individuals assigned to pregabalin with a creatinine clearance (CrCl) of > 60 mL/min received 200 mg three times a day (i.e., those individuals assigned to pregabalin with a CrCl of 30 to 60 mL/min continued to receive 100 mg three times a day). The primary efficacy measurement was pain reduction as reported from an 11-point numeric pain-rating scale (0=no pain; 10=worst possible pain) with values recorded in a daily diary. Secondary endpoints were SF-MPQ questionnaire responses, daily sleep interference scores, Medical Outcomes Study (MOS) Sleep Scale, SF-36 Health Survey, POMS, PGIC, and CGIC. The mean change in the pain score was -1.69 with pregabalin treatment (95% CI, -2.33, -1.05; p = 0.0001) compared to placebo. Secondary endpoints demonstrated a significant difference in favor of pregabalin treatment in SF-MPQ (sensory, affective, and total pain scores), mean sleep interference scores, MOS Sleep Scale, SF-36 (bodily pain and general health perception scales), PGIC, and CGIC. A 50% or greater reduction in pain, compared with baseline, was achieved in 50% of patients in the pregabalin group and 20% in the placebo group (p=0.001). Somnolence was the most common adverse event that led to the discontinuation of pregabalin (11.2%, n=10). The authors concluded that pregabalin is safe and efficacious in the treatment of PHN. Additionally, the results suggested that pregabalin provided a clinically significant, rapid, and durable pain relief without the requirement of a slow and lengthy dose titration.
Partial Seizures
The management of patients with epilepsy is focused on three main goals: controlling seizures, avoiding side effects of treatment, and maintaining or restoring quality of life.23,24 Partial seizures begin in one hemisphere of the brain and produce an asymmetric clinical manifestation, unless they become secondarily generalized.25 Partial seizures have varying degrees of complexity with classifications ranging from simple partial to complex partial seizures. Symptoms vary with severity and may require multiple medications for control. Simple partial seizures include both visible manifestations, such as jerking of a limb and subjective experiences described as an aura. Complex partial seizures can manifest as clouding of consciousness, staring, and repetitive motor behaviors.26 Individuals experiencing complex partial seizures typically do not remember experiencing the event.24 Clinical studies have demonstrated a significant decrease in seizure frequency from baseline with the use of pregabalin.27
French and colleagues performed a multicenter, randomized, double-blind, parallel-group study to evaluate a dose-response relationship and tolerability without titration of pregabalin and placebo (n=455).28 There was an 8-week baseline (patients were eligible for inclusion if they had experienced at least three observable partial seizures in the month prior to screening and six partial seizures in the 8 weeks between screening and baseline with no 4-week seizure-free period) and a 12-week double-blind phase. The patients (age range of 12 to 70 years) had to be receiving one to three antiepileptic drugs (AEDs) with the seizures refractory to at least two AEDs at maximally tolerated doses. Those that met inclusion criteria received 12 weeks of pregabalin 50-, 150-, 300-, 600-mg, or placebo daily divided into twice a day dosing with no dose titration. The median baseline seizure rate was ten per month. Approximately 50% of patients were on two concomitant AEDs and 20% were on three AEDs, with the two most common being carbamazepine and phenytoin. The primary efficacy outcome was a reduction in seizure frequency as measured by the RRatio. The RRatio is a relative measure of change defined as the patient's baseline seizure frequency and the patient's seizure frequency during treatment. The equation is [(T-B)/(T+B)] x 100 with B representing baseline seizure frequency and T representing seizure frequency during treatment. The RRatio is distributed within the range -100 to +100 (+33= 50% increase in seizure frequency; 0= no change; -33= 50% reduction in seizure frequency; -100= complete elimination). In the intent-to-treat analysis (n=453), the seizure reduction for all partial seizures was lower in the pregabalin-treated patients compared to placebo (p<0.0001). Furthermore, there was a percentage reduction in seizure frequency between baseline and endpoint of 54% (RRatio: -37), 44% (-28), 34% (-21), 12% (-6), and 7% (-4) for pregabalin 600-, 300-, 150-, 50-mg daily and placebo groups, respectively. The > 50% reduction in seizures (i.e., responder rate) was 51%, 40%, 31%, 15%, and 14% for pregabalin 600-, 300-, 150-, 50-mg per day and placebo groups, respectively. Doses of greater than 150 mg per day of pregabalin were associated with greater reduction in seizures (p< 0.0001) and greater responder rates (p<0.006) compared with placebo-treated patients. The two most frequently reported adverse events were somnolence and dizziness in the pregabalin-treated patients. There was a dose-related increase in the incidence of discontinuation due to adverse events in the 300- and 600-mg per day pregabalin groups compared to the placebo group; however, the incidence was similar in the 50- and 150-mg per day pregabalin groups compared to the placebo group. The authors concluded that pregabalin is safe and effective for the treatment of partial seizures. Additionally, there is a linear relationship between dose and efficacy for doses of 150-, 300-, and 600-mg per day, but pregabalin 50 mg per day is not different from placebo and appears to be an ineffective dose. Finally, pregabalin appears to be safe and well-tolerated for treatment of partial seizures, and the reported adverse effects are predominantly dose-related.
Treatment of Refractory Neuropathic Pain Patients
Three poster presentations have reported findings of using pregabalin in patients with refractory DPN, PHN, and fibromyalgia syndrome (FMS).
Dworkin and colleagues, in a 12-month open-label study, evaluated the long-term safety and analgesic effectiveness of pregabalin in 106 patients with refractory DPN, PHN, and FMS.29 Study participants must have been enrolled in a double-blind, placebo-controlled pregabalin trial and had inadequate pain relief or intolerable adverse events during 2 weeks of each of the following: tricyclic antidepressant (>75 mg), gabapentin (>1800 mg), and a third-line neuropathic pain treatment (e.g., antiepileptic drugs, opioids, selective serotonin reuptake inhibitors, or selective serotonin-norepinephrine reuptake inhibitors). The dose of pregabalin ranged from 150 to 600 mg/day (divided three times a day). Subjects were permitted to remain on pain relieving medications including gabapentin. Patients were evaluated quarterly after a pregabalin holiday (mean duration 7 days) to evaluate pain using a 5-point scale (0=not at all, 4=very much worse). Only patients with scores > 3 (defined as a relapse) resumed pregabalin treatment. At baseline, all subjects had VAS pain scores of 73, 74, and 75 for DPN, PHN, and FMS, respectively. At the end of the study, DPN, PHN, and FMS patients reported improved VAS pain scores which were 47, 55, and 48, for DPN, PHN, and FMS, respectively. The most common adverse events experienced by pregabalin-treated patients were dizziness and somnolence; moreover, eleven patients discontinued treatment due to adverse events.30
Durso De Cruz and colleagues reported results from an additional 3-month treatment period (i.e., total of 15 months) with the same DPN and PHN patients as above.31 In addition to pregabalin, most patients continued to take a minimum of one previously prescribed pain medication. Mean baseline VAS pain scores for DPN and PHN patients were 72.6 and 74.5, respectively. At the end of the 15 months, the mean VAS pain scores were reduced to 47.2 and 51.3 for DPN and PHN patients, respectively. An analysis was also performed to determine the proportion of individuals with > 30% and > 50% reduction in pain (classified as > 30% and > 50% responders) at 3- and 15-months. For DPN patients, at 15 months 54.6% and 36.4% were classified as > 30% and > 50% responders, respectively. For PHN patients, at 15 months, 41.2% and 32.4% were classified as > 30% and > 50% responders. Pregabalin holidays were consistent in returning the VAS pain scores to baseline during this 15 month study. The overall mean daily doses for pregabalin were 511- and 445-mg for DPN and PHN subjects, respectively. Most adverse events were considered mild-to-moderate in intensity with dizziness, somnolence, and peripheral edema being reported most frequently.
Part II will appear in the November/December 2005 issue of Pharmacotherapy Update along with the references for both Part I and Part II.
Return to Pharmacotherapy Update Index